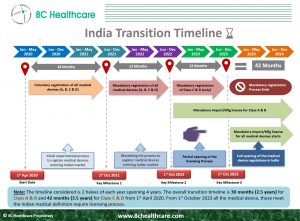
Starting October 1, 2023, 𝒂𝒍𝒍 𝒏𝒐𝒏-𝒏𝒐𝒕𝒊𝒇𝒊𝒆𝒅 𝑪𝒍𝒂𝒔𝒔 𝑪 𝒂𝒏𝒅 𝑪𝒍𝒂𝒔𝒔 𝑫 𝑴𝒆𝒅𝒊𝒄𝒂𝒍 𝒅𝒆𝒗𝒊𝒄𝒆𝒔 shall be subject to the requirements of the 𝑴𝒆𝒅𝒊𝒄𝒂𝒍 𝑫𝒆𝒗𝒊𝒄𝒆 𝑹𝒖𝒍𝒆𝒔 2017.
Since October 1, 2022, all non-notified Class A and Class B medical devices are subject to the 𝑴𝒆𝒅𝒊𝒄𝒂𝒍 𝑫𝒆𝒗𝒊𝒄𝒆 𝑹𝒖𝒍𝒆𝒔 2017 and the manufacturers, whether local or foreign, are required to adhere to the rules for #importing/marketing their devices in #India.
The DCG(I)., while speaking at the 9th International Pharmaceutical exhibition (iPhex 2023) in Hyderabad, confirmed that the regulations are aimed at ensuring quality control and providing a facilitating environment.
As an emerging sector, medical devices manufacturing is on the rise, spurred by an upsurge in demand for many products during the pandemic and the government’s emphasis on ‘#makeinindia‘.
Earlier this year, the #cdsco had issued a circular setting the 𝗢𝗰𝘁𝗼𝗯𝗲𝗿 𝟭 𝗱𝗲𝗮𝗱𝗹𝗶𝗻𝗲 𝗳𝗼𝗿 𝗖𝗹𝗮𝘀𝘀 𝗖 𝗮𝗻𝗱 𝗖𝗹𝗮𝘀𝘀 𝗗 𝗻𝗼𝗻-𝗻𝗼𝘁𝗶𝗳𝗶𝗲𝗱 𝗺𝗲𝗱𝗶𝗰𝗮𝗹 𝗱𝗲𝘃𝗶𝗰𝗲𝘀 to transition from the existing mandatory registration regime to a licensing regime.
It is suggested that the manufacturers/importers apply to the #cdsco with all the requisite documents for 𝗴𝗿𝗮𝗻𝘁 𝗼𝗳 𝗺𝗮𝗻𝘂𝗳𝗮𝗰𝘁𝘂𝗿𝗶𝗻𝗴/𝗶𝗺𝗽𝗼𝗿𝘁 𝗹𝗶𝗰𝗲𝗻𝘀𝗲 at the earliest. Medical Devices that are not licensed after October 1, 2023, may not be allowed for Manufacturing/Import or sale in India.
For more information on the procedure to make the application and documents required for obtaining a manufacturing/import license, you can reach out to contact@8chealthcare.com.